ALMAG ACTIVEis intended to provide physiotherapeutic treatment of pain as well as recovery and rehabilitation measures using a low-frequency pulsed magnetic field, either in medical facilities or at home, upon the recommendation of a physical medicine and rehabilitation physician.
ALMAG ACTIVE enables to:
- Reduce inflammation of joints, increase blood flow & circulation and improve cell metabolism;
- Relieve exacerbation of pain associated with musculoskeletal system disorders;
- Relieve joint stiffness caused by rheumatoid arthritis;
- Increase freedom of movement; improve joint function;
- Promote restore of bones and joints.
Can be used as an essential part of the complex treatment and as a self-acting device.
ALMAG ACTIVE ensures enhanced treatment of musculoskeletal disorders and implies the disease-centered approach.
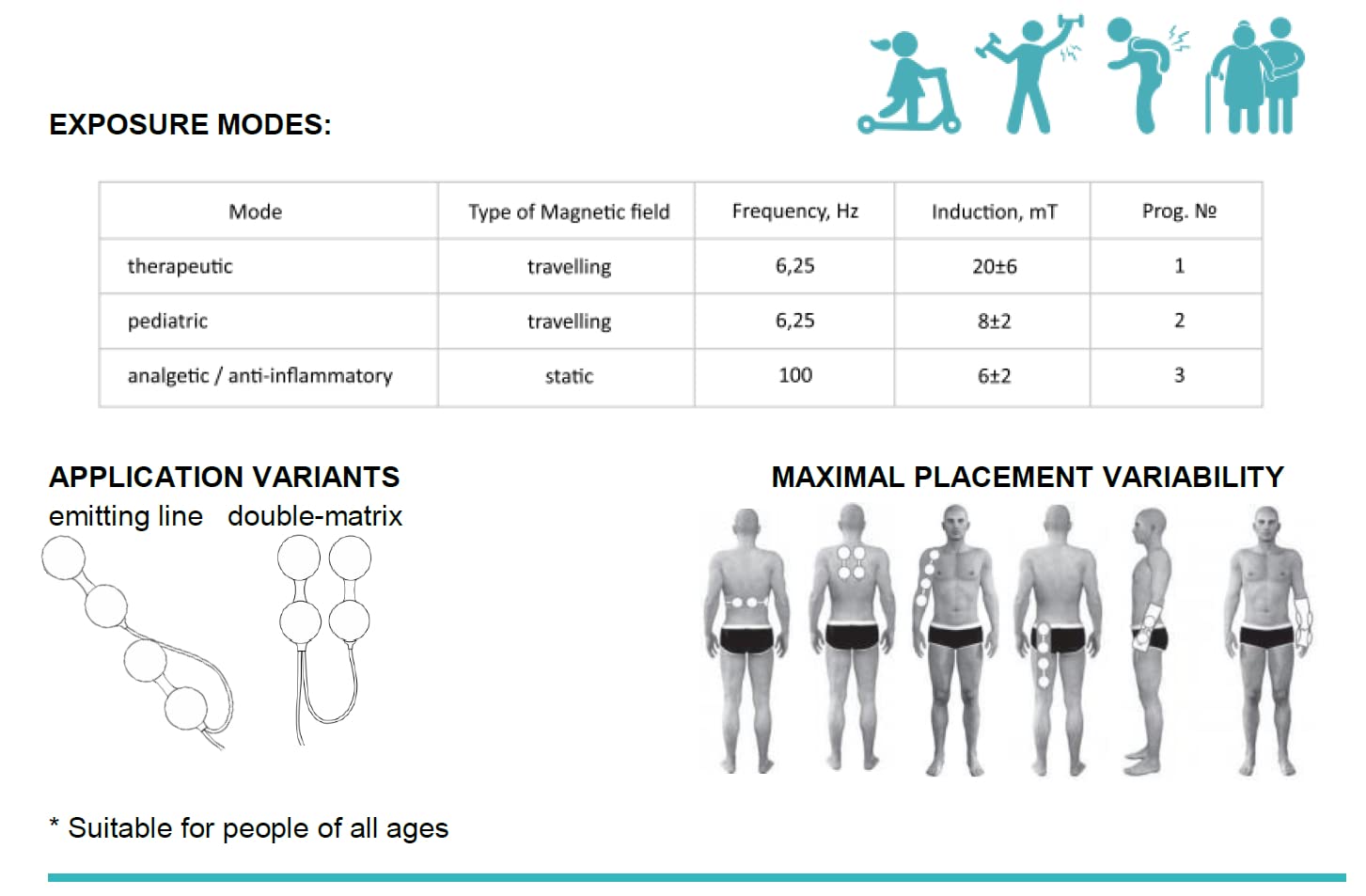
ALMAG ACTIVE can operate using three different modes:
- Analgesic mode: acute pain back and joint relief in the very first disease manifestation of the musculoskeletal system disorders and injures (sprains, fractures , dislocations and others) or in the chronic phrase of the diseases;
- Basic mode: treatment of a disease, thoroughly, by affecting on the core source of the pain ;
- Pediatric mode: treatment of children one month of age and older.
Advanced design and configuration:
- Inductor coils are combined into two groups of two coils per group. The groups may be configured in the form of 2×2 matrix and/or «spline» composed of four inductor coils so that you can ensure the targeted therapy for the pain management in a damaged zone of the body
- The device can heal two / paired joints simultaneously.
How it works:
The device consists of the control unit (current pulse generator) and an emitter comprising four interconnected inductor coils used to provide exposure to individual parts of the body.
Contraindications for use of ALMAG ACTIVE
- Acute infectious diseases and purulent-inflammatory processes of any location;;
- Pregnancy;
- Diseases and conditions accompanied by impaired hemostasis of the hypo- coagulation type (decreased blood coagulation), risk of bleeding and hemorrhagic syndrome, including systemic blood diseases (hemophilia, thrombotic thrombocy- topenic purpura, etc.), as well as bleeding of any genesis and location;
- Malignancies*;
- Conditions preventing from procedure performing: alcohol and drug intoxica- tion, psychomotor agitation of any genesis;
- Uncompensated arterial hypertension of Class 3, arterial hypertension crisis of any class;
- Arterial hypotension (SBP < 90 mm Hg, DBP < 65 mm Hg);
- Aneurysms of aorta and large vessels;
- Hemodynamically significant cardiac arrhythmias and/or intracardiac conduc- tion disturbances (atrial fibrillation and flutter, paroxysmal supraventricular and ven- tricular tachycardia, ventricular extrasystole of degree four or five according to Lown- Wolf-Ryan scale, atrioventricular block degree two or three, sinus bradyarrhythmia);
- Any conditions and diseases of the thyroid gland, accompanied by hyperpro- duction of thyroid hormones;
- Active tuberculosis process of any location;
- Acute and subacute stage of myocardial infarction;
- Implanted cardiostimulator present in the exposure area;
- Diseases and syndromes associated with hypoglycemia;
- Under 4 years of age (insufficient clinical data).
Metal inclusions, if present in bone tissue, is not a contraindication to administration of the device in therapeutic doses.
Metal dental crowns, if present in mouth cavity, is not a contraindication to ad- ministration of the device in therapeutic doses.
NOTE! *Treatment using the ALMAG Active device may be carried out for patients with the diagnosis of a malignant neoplasm only as directed and under the supervision of the attending physician, after a comprehensive antitumor therapy (surgical treatment, chemotherapy course, radiation therapy) and in the absence of metastasis and progression of the tumor process.
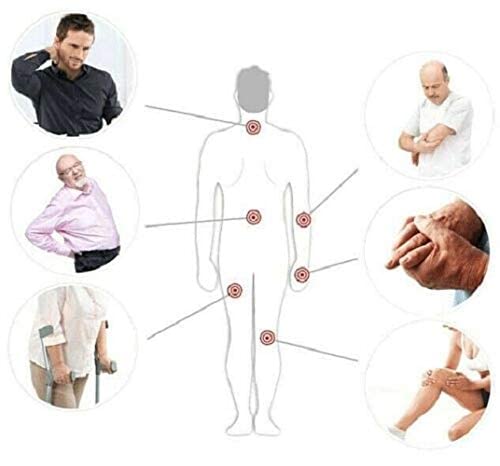
Indications for use of ALMAG ACTIVE
Musculoskeletal system diseases:
- Osteochondrosis;
- Humeroscapular periarthrosis;
- Osteoarthritis;
- Epicondylitis.
Injuries and their after-effects:
- Internal joint injuries;
- Posttraumtic edema;
- Skeletal injuries (bone fractures, dislocations, damage to the ligamentous apparatus – sprains, tears; damage to skeletal muscles, soft tissue injuries);
- Vertebral column and spinal cord traumas.
Diseases of peripheral nervous system:
- Neuritis;
- Facial nerve neurtis;
- Radial nerve neuritis;
- Ulnar nerve neuritis;
- Median nerve neuritis;
- Sciatic nerve (ischias) neuritis;
- Peroneal nerve neuritis;
- Plexitis;
- Neuralgia;
- Trigeminal neuralgia;
- Occipital neuralgia;
- Intercostal neuralgia.
Complications of type I and II diabetes mellitus:
- Diabetic angiopathy of the upper and lower extremities;
- Diabetic polyneuropathy of the upper and lower extremities.
Diseases of venous system:
- Deep vein thrombosis of the lower leg;
- Chronic thrombophlebitis;
- Varicose veins.
Identical medical devices:
- Magnetotherapy device, Medical device registration certificate No. RZN 2017/6194 dated June 20, 2019, Eurasian Economic Union registration certificate No. MI-RUBYKGKZ-000003 dated July 15, 2020, manufactured by JSC Yelatma Instrument Making Enterprise.
Equivalent (analogous) medical devices:
- Pulsed electromagnetic field therapy (PEMF) device, small-sized, Medical device registration certificate No. FSR 2007/00136 dated December 22, 2017, manufactured by JSC Yelatma Instrument Making Enterprise;
- Pulsed Electromagnetic Field Therapy Device «ALMAG-02», Certificate (EU-Certificate) No. 44 232 117836 dated 2019-09-28, manufactured by JSC Yelatma Instrument Making Enterprise;
- Magnetotherapy device, Medical device registration certificate No. FSR 2009/04790 dated November 08, 2016, manufactured by JSC Yelatma Instrument Making Enterprise;
- Magnetotherapy device «Полимаг-02», Medical device registration certificate No. FSR 2011/11993 dated December 20, 2016, manufactured by JSC Yelatma Instrument Making Enterprise;
- Magnetotherapy device, Medical device registration certificate No. RZN 2017/6315 dated November 03, 2016, manufactured by JSC Yelatma Instrument Making Enterprise.
Almag Active has been designed to improve the well-being of patients in an extremely convenient manner. Thus, the device boasts a lot of unique features:
-
-
- an easy-to-use control unit;
- several stimulation options with travelling and stationary charges;
- 3 modes of operation, including an infant mode and the one for enhanced anti-inflammatory effects;
- many coil configurations that can be used for paired organs, among others;
- saved treatment parameters, which makes the following sessions easier;
- sound and light indicators accompanying every session;
- special fasteners to make sure the device is placed securely.
-
The Almag Active device consists of a pulse generator (control unit) and a 4-coil charge emitting system. Those coils are paired together using special fasteners and straps to form 2 sets (each set is made up of 2 coils) intended to be placed on a damaged part of the body. Once a treatment mode is selected on the control unit, the Start button should be pressed so that the device starts emitting magnetic pulses, penetrating tissues.
This low-field stimulation can improve blood circulation, speed up recovery, reduce inflammation, relieve pain, detoxify the body, and help with many musculoskeletal diseases. The effects of the Almag+ treatment largely depend on the selected operation mode and severity of the condition. So, it’s recommended that you consult your physiotherapist to determine the most suitable mode for your case.
Before receiving or administering the treatment with Almag Plus, be sure to read the Operation Manual that comes with the device. You will find more details on the recommended session duration, the range of frequencies, age-specific considerations, and treatment
The device consists of the control unit (current pulse generator) and an emitter comprising four interconnected inductor coils used to provide exposure to individual parts of the body. requirements in that document.





















There are no reviews yet.